Menu

What is Activated Carbon?
Special thanks to Associate
Prof. Dr. Ahmet UYANIK from the Department of Chemistry of ONDOKUZ MAYIS UNIVERSITY
ACTIVATED CARBON
The activated carbons with extremely porous structure are the most important ones of the currently used adsorbents amongst the commonly used industrial adsorbents in order to control environmental pollution. Commercial activated carbons are derived by the activation of processed carbons which are produced by charcoal, peat, lignite, coal, bone, coconut shell, brown rice, nutshell and oil products.
In the early years of 20th century, the patents underlying the current activated carbon production were issued. These patents describe two main principles of activated carbon production which are currently used even today. These principles are chemical activation and gas activation. After 1920, activated charcoal was used in water treatment for the first time but, it could not become widespread. Because the odor of chlorophenol in potable water of Germany caused big problem in 1927, usage of the activated carbon during the preparation of city water have become crucial. The activated carbon was utilized in granular form in Hamm Water Works in 1929 and apart from this, in Michigan Bay City in 1930 by Harrison and in 1929 as a powdered form in order to remove odors from potable water by Spalding. As of 1932, 400 factories were utilizing activated carbon for controlling the unpleasant odors. This figure increased by 1200 factories in 1943.
General Characteristics of Activated Carbon
Activated carbon is a generic term used for defining the carbonized adsorbents due to its big crystal form and extremely large inner pore structure. The activated carbons have extremely high porosity and inner surface area as well as being useful and non-hazardous products on human health (5). The activated carbons are named as adsorbents simply because they can absorb the molecules and ions within a solution into the inner surfaces through their pores.
Surface Area
Inner surface of an activated carbon (activated surface) is commonly defined as BET surface (m2/g). Surface area is measured by means of nitrogen gas (N2). Inner surface area of the activated carbon particles used for water treatment should be requested to be almost 1000 m2/g. Since the polluting substances are retained on the surface of the activated carbon, the size of the surface area is an effective factor to eliminate pollutions. In principle, it is thought that the larger surface area means greater adsorption centers. Numerical values regarding the surface area and pore system of the activated carbon which are available in the literature are as follows;
Numerical values regarding the surface area and pore system of the activated carbon.
Surface Area 400-1600 m2/g (BET N2)
Pore volume: >30 m3/100g
Pore width: 0,3 nm-1000 nm
The surface of carbon particle attracts the gas, liquid and solid materials in and forms a thin film layer on the surface area, in other words it absorbs them. There are two main reasons why activated carbon is preferred as adsorbent:
1. It has an absorbing surface to absorb certain substances,
2. It has a large surface area to retain too many substances.
Pore Size
Another parameter which has a significant effect on the elimination of the pollution is pore size. The determination of pore size is very useful method for identifying the characteristics of carbon. The pores may be cylindrical and conical-shaped. The following photograph is taken by Scanning Electron Microscope (SEM) and shows the pore structure of activated carbon.
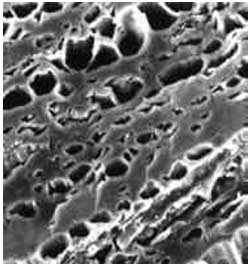
Figure 1: The pore structure of activated carbon. Taken by SEM.
Pore structure is an important element more than total inner surface area for the process of adsorption. The sizes of pores should align with the particle diameter of pollutants to be removed. This is because the attraction force between the carbon and adsorbed molecules is greater than the molecules that almost have the same size with pores.
The International Union of Pure and Applied Chemistry (IUPAC) has divided the pore size of adsorbents into four categories according to their radiuses;
1. Macropores(r > 25 nm)
2. Mesopores (1 < r < 25 nm)
3. Micropores (0,4 < r < 1 nm)
4. and submicro pores(r < 0,4 nm) .(Figure 2)
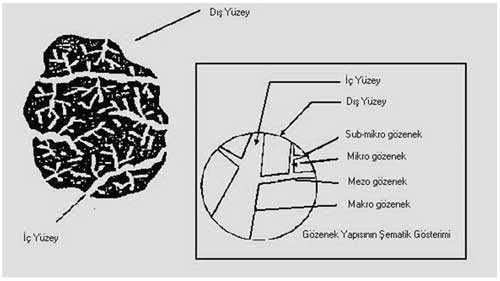
Figure 2: Schematic activated carbon model
Pore structure of activated carbon important for adsorption and desorption is shown schematically in Figure 2. Majority of inner surface consists of micropores (~95%) Macropores are relatively less important for adsorption but they should act as transmitter in order to accelerate the diffusion towards the micropores. (6) While macropores enables the molecule to take in the activated carbon, mesopores transport it to the deeper areas. Meanwhile, micropores are used for adsorption.
Types of Activated Carbon
The best activated carbons used for the wastewater systems are derived from a variety of coals and natural materials. These are: hard coal, charcoal, peat, lignite, coal, bone, Coconut-rice and nut shell, fruit seeds and oil products. The activated carbons derived from those materials have rigid and dense structure in general. They may be used in water for a long time without any degradation. It is possible to produce the activated carbons with different characteristics. These are;
1. Powdered activated carbons,
2. Granular activated carbons,
3. Activated carbons in pellet form.
The powdered activated carbons are derived from the chemical activation of carbon. These carbons are commonly used for the treatment of wastewater. Granular products and pellets produced through the gas activation are commonly used for the purification of gases. However, it is stated that activated carbons in granular form give satisfactory results in wastewater treatment systems. The granular and powdered activated carbons produce spectacular outcomes in the removal of organic and inorganic substances. These activated carbons have been used for cleaning the wastewater subjected to biological treatment and the wastewater consisting of organic-based industrial wastes for many years.
Activation Techniques
All types of carbon-rich substances may be used for the production of the activated carbon by activating them through different methods. These activation methods are divided into two categories as chemical activation and gas activation.
Chemical Activation
This technique is generally used for the activation of raw materials derived from peat and charcoal. Raw material is saturated with zinc chloride, phosphoric acid or potassium hydroxide. After that, it is heated up to 500-800 °C to activate the carbon. The activated carbon is washed out, dried and then crushed into powder. The activated carbons generated by chemical activation are commonly used for the adsorption of macromolecules and they have extremely large pores.
Gas Activation
This activation technique is generally used for the activation of coal and peels. Raw material is firstly subjected to a thermal treatment called carbonization. This process helps to produce a carbonaceous product with small pores. And then, the activation process takes place in an inert gas atmosphere and at a temperature range of 800-1100°C . By doing so, the intermediate material generated by the carbonization process at the beginning is converted into gas-phase by means of the following water-gas reaction and so the quantity and size of the existing pores are augmented accordingly.
C + H20 ® CO + H2 -175,440 kJ/(kg mol)
This is an endothermic reaction and the required heat for reaction is protected by burning of partially formed CO and H2.
2CO + O2 ® 2CO2 + 393,790 kJ/(kg mol)
2H2 + O2 ® 2H2O + 396,650 kJ/(kg mol)
The derived activated carbon is classified and made ready for use after being screened and dedusted. Activated carbons generated by gas activation have sound pore structure like the ones generated by chemical activation. They are used effectively for the adsorption of molecules and ions both from liquid and gas phases.
ADSORPTION
Adsorption is defined as the accumulation of a substance on a certain interface or intersection and increase in its concentration(7). The said interface may be the contact surface between the liquid and gas or the solid and other liquid substance. In other words, an adsorption is the adhesion of molecules onto the surface due to the forces of attacking to surface. Adsorption process of a dissolved compound by activated carbon has three steps ;
1. Transmission of adsorbed substance to the outer surface of adsorbent,
2. Diffusion of adsorbed substance to the pores of carbon except for little amount of adsorption occurred at the outer surface,
3. Adsorption of solution in the inner surfaces of adsorbent.
According to another source, an adsorption occurs at three basic steps: These are;
a) Film diffusion: Solute molecules - to be adsorbed - form surface film by incorporating into the carbon particles.
b) Pore diffusion: Transmission of solute molecules from carbon pores to the center of adsorption.
c) Adhesion of Solute Molecules to Carbon Surfaces. An adsorption occurs when solute molecules stick onto surface area.
Types of Adsorption
It is indicated that adsorption on the activated carbon may occur at three different processes (9).
Physical Adsorption
If adsorption occurs by means of unbalanced Van Der Waals forces, it is defined as physical adsorption. This type of adsorption is reversible in terms of thermodynamic process. It is characterized by low adsorption heat and the degree of adsorption is inversely proportional to temperature.
Chemical Adsorption
It stems from the creation of chemical bond with the monomolecular layer of adsorbed substance on the surface due to the valence forces of surface molecules. Adsorption requires high temperature and is not reversible in terms of thermodynamical aspect. If temperature rises extremely, physical adsorption may transform into chemical adsorption (10).
Electrostatic Adsorption
It is defined as the effect of electrical attractive forces which are responsible for the adsorption of solutions onto the activated carbon. Electrical attraction between negatively-charged carbon particles and positively-charged adsorption molecules or ions eliminates the negative effects occurring at the time of diffusion, thus increasing the effectiveness of adsorption.
Recovery of Adsorbent
As molecules are adsorbed onto the adsorbent surface, there is much more little space for the adsorption of new molecules. As a consequence, the adsorbent loses its effective adsorption property. The act of getting the effective adsorption property of adsorbent is called as "recovery". Physical force of activated carbon should be adequate to resist during the recovery period. However, due to the reasons of thermal diffusion, shrinkage and fission of structure at the end of the process, a little activated carbon is lost and oxidized.
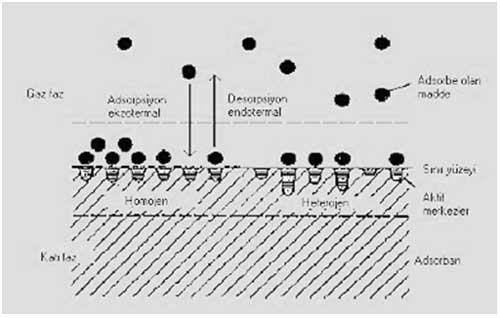
Adsorption - Desorption Processes on Solid Phase
Adsorption, as well as the other definitions, is enrichment of the substances which are dissolved by means of adsorbing on the relevant surface of a solid adsorbent. The bond forces of the atoms on the surface of adsorbent named as active center are not fully saturated. Adsorption of foreign molecules occurs in these active centers. A substance adsorbed on an adsorbent is replaced by a substance that is more heavily adsorbed in proportion to itself. Replaced substance is either desorbed or released by carbon. This process proceeds for the absorption process of the mostly preferred types. Chemical adsorption arises from the functional groups of adsorbed substances and it is interacted with adsorbent to create a stable bond. Desorption event is more suitable for physically adsorbed substances than chemically adsorbed substances. Adsorption and desorption processes on solid phase are shown schematically in Figure 3.